I got 110 copper bar delivered next day to Phoenix from them. I didn't ask or pay for next day, that's just how it worked.Their inventory is insane - I got Titanium bolts from them delivered same day (only in LA area)
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Copper washers and brass bolts or screws for battery terminals
- Thread starter sid the seagull
- Start date
Hedges
I See Electromagnetic Fields!
- Joined
- Mar 28, 2020
- Messages
- 20,507
Stainless steel into Aluminum is not great. It can cause galling.
If turned under load?
Securing a stud with loctite shouldn't have that problem.
I am going to just use all brass hardware with and side step the issue entirely. Hardware has been ordered.If turned under load?
Securing a stud with loctite shouldn't have that problem.
Factory400
Solar Enthusiast
- Joined
- Jul 15, 2020
- Messages
- 387
I am going to stick with stainless fastening hardware to maximize joint pressure and solid machined copper bus bars.
Careful about torque, so you don't strip the threads in the battery.I am going to stick with stainless fastening hardware to maximize joint pressure and solid machined copper bus bars.
Factory400
Solar Enthusiast
- Joined
- Jul 15, 2020
- Messages
- 387
Definitely - I am worried about that for sure.Careful about torque, so you don't strip the threads in the battery.
I am fortunate enough to have the tools to repair a damaged thread if I go too far.
Bob B
Emperor Of Solar
- Joined
- Sep 21, 2019
- Messages
- 8,604
John knows.
The terminal-buss bar connection should be carrying most of the current, not the fastener that holds it together.
Galvanic compatibility is more important, which brass and aluminum (battery terminal) are not. The brass will cause the aluminum to corrode
View attachment 32683
From that chart, it looks like stainless is the worst ... and that is what most of us have been using. Brass is in the middle and may be a good compromise between the hardness and the galvanic response.
Actually, it looks like the cheapest option .... zinc coated steel might be the best option.
Factory400
Solar Enthusiast
- Joined
- Jul 15, 2020
- Messages
- 387
I have been putting stainless STI type inserts into aluminum and using stainless fasteners in life-critical aerospace applications for a very long time. Admittedly, I never really researched the chemistry at all, but the engineers that specified these fasteners did. My particular cells have stainless inserts in the terminals which is far stronger than the bare aluminum in axial strength and also allows more mating cycles. Not sure if this is common or if I am just lucky.From that chart, it looks like stainless is the worst ... and that is what most of us have been using. Brass is in the middle and may be a good compromise between the hardness and the galvanic response.
Actually, it looks like the cheapest option .... zinc coated steel might be the best option.
So far, I have never seen any corrosion even in significantly corrosive environments. Stainless seems unlikely to be a problem in this application.
Now you tell me. I do still have stainless steel grub hubs. I wish you guys would make up your minds. I am just a poor EE. I skated through this stuff in school.John knows.
The terminal-buss bar connection should be carrying most of the current, not the fastener that holds it together.
Galvanic compatibility is more important, which brass and aluminum (battery terminal) are not. The brass will cause the aluminum to corrode
View attachment 32683
Read this.Definitely - I am worried about that for sure.
I am fortunate enough to have the tools to repair a damaged thread if I go too far.

Source of Belleville washers for 300kgf fixture?
Cool, If you bought some send me a couple (4) and I will test them. I was hoping to follow your lead ;).
I meant this one.

 diysolarforum.com
diysolarforum.com
I got 62 lb compression force with a 5/16" bolt and a finger tight nut.

Source of Belleville washers for 300kgf fixture?
Cool, If you bought some send me a couple (4) and I will test them. I was hoping to follow your lead ;).
I got 62 lb compression force with a 5/16" bolt and a finger tight nut.
Bob B
Emperor Of Solar
- Joined
- Sep 21, 2019
- Messages
- 8,604
I can't argue with your experience since even though I have an extensive background in electronics ....I am new to these type of factors that come into play with a DIY battery pack.I have been putting stainless STI type inserts into aluminum and using stainless fasteners in life-critical aerospace applications for a very long time. Admittedly, I never really researched the chemistry at all, but the engineers that specified these fasteners did. My particular cells have stainless inserts in the terminals which is far stronger than the bare aluminum in axial strength and also allows more mating cycles. Not sure if this is common or if I am just lucky.
So far, I have never seen any corrosion even in significantly corrosive environments. Stainless seems unlikely to be a problem in this application.
I am currently using stainless grub screws that can be held with a hex wrench while tightening the flange nut down on the bus bar. I am happy that this takes the twisting stress off the aluminum threads in the cell. I haven't used any of the thread lock that some have been using ... still trying to decide whether to keep this for my final setup.
I have noticed that if I remove the nut from the grub screw .... and then start removing the grub screw from the cell, it initially takes quite a bit of force to start the grub screw moving and I hear a "click" and the screw releases from the aluminum threads and comes out easy the rest of the way. I'm not sure what is causing the grub screw to seize a little with the aluminum thread.
This was after only having the pack put together for a couple of weeks.
Galvanic corrosion is different than what you are thinking. Aluminum oxide is a very poor conductor, your screws and bolts are not carrying the current, the surface where the busbar and the battery terminal meet carries the current. Galvanic corrosion is the chemical reaction when two dissimilar metals meet, basically any corrosion that would have occurred, will only occur on one of the metals. Basically it means the aluminium will corrode first and at a much higher rate. Aluminum oxide is the result, and it is terrible for an electrical connection, much worse than copper oxide. Just wipe some of the many and cheap compounds specifically designed for copper to aluminium on the terminal face, and the busbar. The stud and nut, don't worry about, they don't conduct current anyway. Basically you just put a very (and I stress very) thin layer on the terminal face and the part of the busbar to exclude oxygen and thus corrosion, the nice tight interface between the battery terminal and the busbar will squeeze and exclude oxygen and corrosion. Unless you get a lot of salt spray into the battery compartment, no problem. I doubt the corrosion of the screws will be a problem until long after the battery has reached end of life. It might weaken the mechanical connection given enough time, but we are talking decades in any environment I would put a battery.Now you tell me. I do still have stainless steel grub hubs. I wish you guys would make up your minds. I am just a poor EE. I skated through this stuff in school.
Aaron_85
New Member
You also must take into consideration the busbar-fastner compatibility as well, but the aluminum post on the cell being the hardest to replace, is the most important to worry about not ruining  The paste is very important at stopping the oxygen getting in and starting the process.
The paste is very important at stopping the oxygen getting in and starting the process.
you also reference charts like this, and the closer the metals are on the chart the lesser the effect is.
Maybe we need to start talking about using galvinised/zinc plated fastening hardware instead of stainless in/on our battery systems?
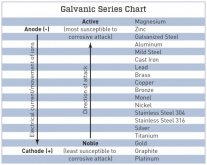
you also reference charts like this, and the closer the metals are on the chart the lesser the effect is.
Maybe we need to start talking about using galvinised/zinc plated fastening hardware instead of stainless in/on our battery systems?

Hedges
I See Electromagnetic Fields!
- Joined
- Mar 28, 2020
- Messages
- 20,507

Carbon Screws by Schmolke Carbon
Our carbon screws are manufactured in a highly specialised pressure extrusion procedure using only the best quality carbon.
You also must take into consideration the busbar-fastner compatibility as well, but the aluminum post on the cell being the hardest to replace, is the most important to worry about not ruiningThe paste is very important at stopping the oxygen getting in and starting the process.
Maybe we need to start talking about using galvinised/zinc plated fastening hardware instead of stainless in/on our battery systems?
Most, if not all, of the anticorrosion compounds include zinc as a sacrificial element. As the chart you posted indicates, zinc would be the first thing to corrode. I would stick with copper or silver plated copper busbars, and stainless steel grub screws and nuts (either lock nuts, or a stainless lock washer). McMaster Carr has a great selection, very good prices, and fast shipping in my experience. I ordered a 6 foot long 110 copper busbar from them and it arrived next day (LA to Phoenix). It wasn't sent as next day shipping, but UPS was on the ball. Their biggest problem is not telling you how much shipping is until it ships and they charge your card, but the shipping is always very reasonable. I ordered 25mm stainless grub screws, washers and nordlock bolts as well as the busbar (comes with an assay certificate) for like $101. I didn't order the silver plated copper busbar because once cut and drilled, parts are no longer plated. Silver plated is like $10 extra.
My take on the problem: use aluminium studs, nuts and washers, and aluminium busbars. That way you only have the 2 connections to the loads to worry about galvanic corrosion and it's not on the expensive cells but on some cheap and easily replaceable busbars 
Factory400
Solar Enthusiast
- Joined
- Jul 15, 2020
- Messages
- 387
Aluminum studs seem extremely weak for this sort of thing.My take on the problem: use aluminium studs, nuts and washers, and aluminium busbars. That way you only have the 2 connections to the loads to worry about galvanic corrosion and it's not on the expensive cells but on some cheap and easily replaceable busbars
Is galvanic corrosion a real problem that needs to be solved or a rare corner case in specific applications?
Aluminum studs seem extremely weak for this sort of thing.
I see it as an advantage, if you break something it's likely to be the stud instead of the cell terminal
Is galvanic corrosion a real problem that needs to be solved or a rare corner case in specific applications?
It is a real problem. Now, is it a problem here? hard to tell. Also, it depends on a lot of factors, main one being humidity. On a boat it'll definitely be a problem, and in a house in the desert it'll definitely not. For cases inbetween it's complicated.
Personally I'd say if it's in a house in a non-tropical area then it's fine. If it's in a tropical area, or in the basement, or in a vehicule I'd say it would be wise to take precautions against it. But that's only an educated guess, YMMV.
Factory400
Solar Enthusiast
- Joined
- Jul 15, 2020
- Messages
- 387
Interesting point.I see it as an advantage, if you break something it's likely to be the stud instead of the cell terminal
I am kinda thinking of a cracked aluminum stud while the system is under load - the resulting loose connection would become an inadvertent TIG welder with nothing to stop it. (have your cameras ready for the viral YouTube video!)
I will vote for the most mechanically sound fastener possible in this application.


