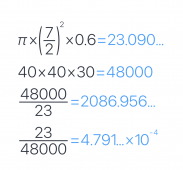Have you measured temperature change and water volume though a heat exchanger? Or rate of temperature change for a volume of water?
That would let you determine Joules or BTU/hour for a given power input and temperature delta between hot/cold sides.
Math...
The secondary side has four main components other than water.
- aluminum heat block touching peltier stack
- aluminum heat block touching process volume
- silicone tubing
- water pump
Rough numbers..
- 176 grams. 200x40x12mm. aluminum 2.7g/cm^3. solid block would be 259.2 grams. implies 30.8 cm^3 volume inside this block.
- 75 grams. 80x40x12mm. aluminum 2.7g/cm^3. solid block would be 103.7 grams. implies 10.6 cm^3 volume inside this block.
- ~40 cm length. inner diameter 7mm. 0.38 cm^2 area inside. implies ~15 cm^3 volume inside tubing.
- just going to guess and say 3 cm^3.
Ok.. so I'm going to
estimate 30.8+10.6+15+3 =
59.4 cubic centimeters of volume in this loop.
Water is ~1 gram per cubic centimeter. 4,184 Joules to increase 1 kilogram of H2O temperature by 1 degree celsius.
4.184 Joules to increase 1 gram of water by 1 degree celsius.
~60 grams of water.
So if I haven't gone totally off the rails with my calculations and estimates...
approximately 250 Joules per degree celsius changed in the water loop.
This is purely an estimate of the water volume, and ignores the specific heat capacity of the aluminum blocks.
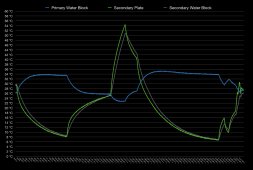
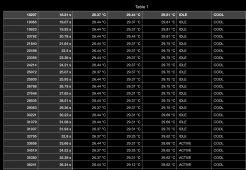
I wrote a script to take the previous 25 log entries and tabulate the average temperature change over that period. That worked out to about 21 seconds of past data being taken into account. Attached is that printout of the derived data showing delta temperature of the secondary aluminum water block over time.
During the period of heating the secondary side, a steady 0.25-0.35 degrees celsius per second was averaged. Since this thread is talking about air heating and water can service that..
That means... 75 Joules per second... 75 W thermal transfer? 255 BTU/hr? Doesn't really seem like every element is running at good efficiency.
edit: mass of aluminum in loop is ~250g and at 0.9 joule / gram Kelvin it should account for an additional 225 Joules per degree change. ? any thermodynamics knowledge advice here would be greatly appreciated.
edit2: there are copper heat spreaders inside the stack.. 250x63x1mm -> ~15.75 cubic cm.. it's 140g... 0.38 J/g K for copper.. there are 5 of these copper plates... that implies an additional 266 Joules per degree Celsius change... probably better to delete those, not clear if they actually provide a benefit.. i included them in the hopes of ensuring the adjacent peltier modules at each layer would stay close in temperature.
My end goal is to individually power each layer, and individually monitor the Seeback Voltage of each string/layer. This would allow for realtime feedback of the delta T per layer... still working on that.. As I move on to 7-10 layer designs, individual power control and monitoring of Seeback Voltage will be essential to efficient operation under a variety of conditions. This test was with 4 layers because i got lazy and got tired of applying thermal pads on the way to 7 layers prototype. It's so tedious to apply the thermal paste that I want to design 3D printed brackets with edge space carved out so I can just add a bunch of thermal paste and put it in a vice and have the excess go into the seam.
I ignored the thermal mass of the water blocks, so I am probably very off the mark by now. But I tried.
this makes me want to add more layers and lower the voltage. in my tests, 1.0V per module resulted in twice as many degrees change per watt as 2.0V per module.......
at this point just writing a script to model the individual surface area, mass, specific heat capacity, instantaneous temperature, of each material in the stack seems like my only real chance of being rigorous.
Anyways, feedback welcome.