Section 4
The First 21P4S Battery
Electrical Circuit Fundamentals and Laws
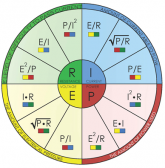
The construction of this 212P4S battery is a massive series/parallel circuit. And always the laws of electricity developed by Geog Ohm and Gustav Kirchhoff will apply. The two laws that every circuit designer should be familiar with are Ohm’s law – E=IxR and the Power law P=IxE. There is a nice pie chart that shows all the forms of these two laws.
The first design variable to establish is the voltage. I have chosen 12 volts for my battery design. At this point, I think it is important to note that saying that I am designing a 12-volt battery will not actually mean that the voltage will be 12 volts. The actual battery voltage will depend on battery chemistry and state of charge. Using a fix number at this point simplifies the conversation.
The next design criterion is the power available from the battery. In battery systems, we rate batteries in amp-hours, Ah, which is the unit that battery manufacturers use. I have decided to build a battery in the 100Ah range, and the actual battery that I am building is rated at 126Ah.
Now if I could get 126 amperes from this battery over a period of 1 hour at a voltage of 12 volts, then the power formula, P=ExI, shows me that the battery would produce 1512 watts for that hour or 1.5 kilowatt hours, or 1.5kwh.
So, will all the internal connections of the battery have to carry the full 126 amps? To answer this question, we must dig deeper into the actual battery structure.
Each of the 32650 LFP cells that I have used is rated at 6Ah. The cells are arranged such that only three cells of the same polarity are directly in the path of current flow. Therefore, each path of current flow will carry 18 amps. Since there are 7 paths of current flow, the 7 paths combine to give the battery 126 amps of current flow.
When we look closer at the cell specifications, we see that the cell can deliver 18 amps continuously, and 60 amps for as much as 10 seconds. What do we do to account for these larger values?
As part of the design, we will be incorporating a Battery Monitoring System, BMS, which can be programmed to give us some current limits. We can design for expected normal currents and use the BMS as a safety device.
Next, we look at the cell to cell connections. Our connections to each cell will be through spot welds. The best material that I have for spot welding is nickel, but nickel has a relatively high resistance. The best, commonly used connection material would be copper, but welding copper is not easy and requires much more heat. In the construction of the battery, we must limit the heat applied to the cells.
The amount of current that a conductor can carry without heating up is directly related to the internal resistance of the conductor. This internal resistance is related to the actual material of the conductor and the cross-sectional area of the conductor.
The heat dissipated in the internal conductors is typically called the loss. This a power loss, as heat, that does not benefit you. This heat can do damage to your battery by directly harming the cells themselves, and the heat can put stress on your welds.
My choice to address my concerns maybe unusual and does take more time to build, but I believe that will address my concerns.
First, I chose 0.3mm by 8mm tape for my direct to cell connections. I have found that I can successfully weld this tape to the cells. I then run parallel 12awg copper wire to carry the bulk of the current.
By getting the bulk of the current into the copper, I also reduce the loss in the battery. Also, by minimizing the heating in the conductor, I have minimized the thermal growth and the strain on the welds.
I first heard of the following initials and phrase from the author Robert Heinlein-TANSTAAFL which translates to ‘There Ain’t No Such Thing As A Free Lunch’. What I have built requires more time and many more welds. Time will tell if my decisions are wise.
 document goes as follows: RV house batteries are important, AGM lead acid batteries are heavy and old technology, commercial LFP batteries have limited physical sizes and capacity options, commercial LFP batteries are expensive, and building a battery to meet my needs is personally satisfying.
document goes as follows: RV house batteries are important, AGM lead acid batteries are heavy and old technology, commercial LFP batteries have limited physical sizes and capacity options, commercial LFP batteries are expensive, and building a battery to meet my needs is personally satisfying. document goes as follows: RV house batteries are important, AGM lead acid batteries are heavy and old technology, commercial LFP batteries have limited physical sizes and capacity options, commercial LFP batteries are expensive, and building a battery to meet my needs is personally satisfying.
document goes as follows: RV house batteries are important, AGM lead acid batteries are heavy and old technology, commercial LFP batteries have limited physical sizes and capacity options, commercial LFP batteries are expensive, and building a battery to meet my needs is personally satisfying.