svetz
Works in theory! Practice? That's something else
It would be interesting to see under a microscope. You've probably seen it, but https://link.springer.com/article/10.1007/s11665-018-3139-7
Actually I hadn't seen that! Thanks for the linkIt would be interesting to see under a microscope. You've probably seen it, but https://link.springer.com/article/10.1007/s11665-018-3139-7


Ok, so long post, but I felt it important to share. Lastly, thank you so much for putting your work out there in a blog and on this forum. I literally refresh many times a day waiting for updates!
I have not ordered TPABr, given that TMPhABr is less symmetric, less aliphatic and more polar I thought it would be a better candidate in terms of having higher solubility while its perbromide - given the melting points between TPABr3, TMPhABr3 and TBABr3 - seems to be the most stable and therefore probably the one with the lowest solubility and highest "sequestering efficiency". In the literature sequestering agents containing aromatic groups tend to do better at capturing more bromine atoms per mole (since they seem to be able to stabilize larger polybromides).I then came across the same Chinese paper you did regarding tribromide confinement with a complexing agent. I ordered myself a Kg of TPABr as used in the paper. I did some back of the envelope calculations as you did and realised the energy density values reported were misleading (par for the course it seems). Even so, I thought it worth experimenting with.
1) Solubility of TPA Br in the presence of Zinc ions
2) Zinc corrosion
For me 10% PEG-200 seems to have solved this issue, at least at the current densities I have used. You can probably go up to 20-30% PEG while still having a working battery if you want to avoid dendrites, although the cost in energy efficiency might be significant.3) Dendrites
At the moment, I'm experimenting with water-in-salt electrolytes, aprotic solvents, thixotropic modification and complexing agents.
I have been testing with graphite felt - 10mm (I got excited and bought a 5m roll from Alibaba) as the cathode. I'm plating directly on to a graphite anode. My standard testing cell is about 30 mm in diameter and 10mm deep (I can set the depth). The graphite felt cathode is joined directly onto another graphite end stop with graphite / nitrocellulose binder.
My main concern is total stored Wh/L (and stability).
Provided your potential stays below 1.9V the hydrogen evolution should be pretty limited, at least this has been my experience using Zn anodes.Regarding corrosion, I actually meant hydrogen evolution. I would be very interested to know if you are having any issues there.
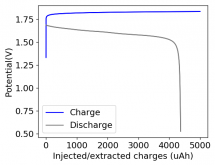
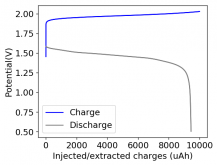
I'll definitely focus on this type of experiment as soon as I get to a value of Wh/L and Wh/kg that seems good enough!PS: Your latest results with a higher charge is impressive! I would love to see discharge curves with a delay included after charging!
Surface resistance as quoted from the manufacturer: Ω/cm2 0.145-0.183. That number seems fairly generous, but I can confirm that the conductivity is very high. The main issue is that it is not activated, so the surface area is low. I don't have a good way to measure it, but based off super-capacitor measurements, its orders of magnitude lower than regular activated carbon. My latest tests have used this felt with a coating of activated carbon.What's the conductivity and surface area of the felt you're using? The GFE-1 graphitic felt I am using has very high conductivity, so I haven't had to use any additional conditioning to improve its characteristics but I'm interested on the properties/cost of your felt.
With the TPABr it is fairly low, but its definitely still there and it will make sealing a cell tricky. I don't think hydrogen recombination is going to help here.Provided your potential stays below 1.9V the hydrogen evolution should be pretty limited, at least this has been my experience using Zn anodes.
Is that based on the electrolyte volume, or the total cell inner volume, including separators etc? So, the 35 Wh/L I achieved was certainly no free lunch! The self discharge was high. I didn't measure it, but at least 50% after 24 hours. There were some dendrites, but this wasn't too much of an issue as the electrode spacing was >4 mm.In terms of Wh/L I have achieved around ~20 Wh/L
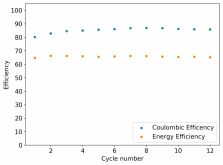
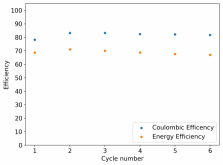
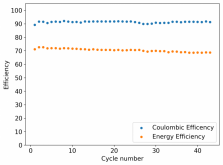
Recombination is certainly not practical. Given that PEG-200 or some other additive is likely to be necessary to avoid dendrites, I think you might want to test this to see how it affects your hydrogen evolution as well. Given my Coulombic efficiency values, I do believe I might be losing some amount of charges to H2 production, especially in cells where my potential reaches close to 2V.With the TPABr it is fairly low, but its definitely still there and it will make sealing a cell tricky. I don't think hydrogen recombination is going to help here.
This is based on total volume of the cell (all materials). If I used only electrolyte volume, it would be like >45Wh/L, but that wouldn't be very indicative of practical energy density values. Using 8 layers of the fiberglass separator I used I'm usually at 2mm of electrode spacing, but I have been doing some tests at 16 layers, so that I can tolerate a bit more of Zinc dendrites.Is that based on the electrolyte volume, or the total cell inner volume, including separators etc? So, the 35 Wh/L I achieved was certainly no free lunch! The self discharge was high. I didn't measure it, but at least 50% after 24 hours. There were some dendrites, but this wasn't too much of an issue as the electrode spacing was >4 mm.