danielfp248
Battery researcher
- Joined
- Sep 7, 2020
- Messages
- 429
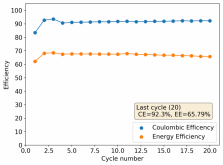
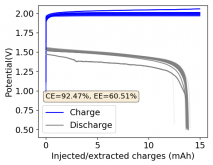
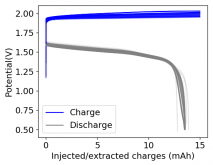
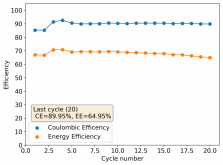
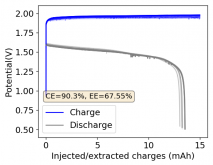
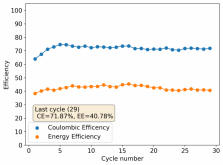
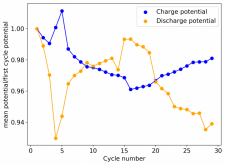
Thanks for your comment! The problem with this design is that the internal resistance is so high - due to the distance between electrodes - that the energy efficiency will be abysmal (probably lower than 10%). Putting 10 Wh in to get 1Wh out is not practical unless you have a lot of energy to waste. In practice if you want to get high energy efficiencies (>50%) you will need to keep your electrode distance below 5mm. This is because of limits in the conductivity of Zinc Bromide solutions. This is why these "jar batteries" are not practical Zn-Br batteries. Note that their Coulombic efficiency can still be quite high (>80%) but this does not mean the battery is any good in real terms.According to Rob on YouTube, this design doesn't or shouldn't have much issues with dendrites problems, please if you have the time and resources do take a look this too
With electrode distances so short, zinc dendrites are sadly going to be a problem we will have to solve.