danielfp248
Battery researcher
- Joined
- Sep 7, 2020
- Messages
- 429
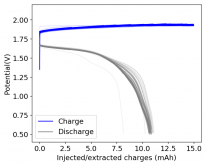
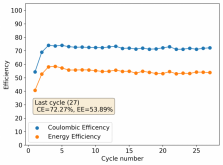
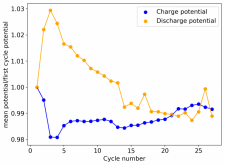
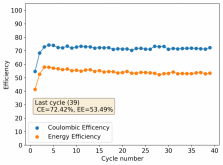
First 29 cycles of an inverted battery (GFE-1 cathode pretreated with 10% TMPhABr on top) using the electrolyte described in #160
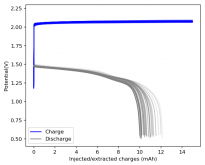
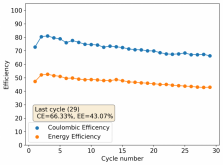
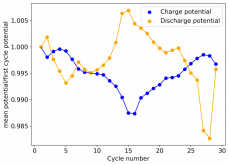
Charge and discharge potentials have remained within 2% of their initial values. I want to run this battery for more than 50 cycles to make sure that it will not die before doing additional experiments. Despite the lower CE and EE values, it does seem that an inverted configuration is the only viable one in a stable ZnBr static battery, due to the need to keep hydrogen from escaping the device.
A normal configuration (cathode at the bottom) makes it hard for hydrogen bubbles at the anode to leave or react, making the surface area of the anode decrease with time. If the bubbles escape, then all that hydrogen evolution makes the electrolyte irreversibly more basic, making the pH go higher and higher with time, eventually completely killing the battery (as Bromine is unstable at higher pH values and ZnO forms at the anode surface).



Charge and discharge potentials have remained within 2% of their initial values. I want to run this battery for more than 50 cycles to make sure that it will not die before doing additional experiments. Despite the lower CE and EE values, it does seem that an inverted configuration is the only viable one in a stable ZnBr static battery, due to the need to keep hydrogen from escaping the device.
A normal configuration (cathode at the bottom) makes it hard for hydrogen bubbles at the anode to leave or react, making the surface area of the anode decrease with time. If the bubbles escape, then all that hydrogen evolution makes the electrolyte irreversibly more basic, making the pH go higher and higher with time, eventually completely killing the battery (as Bromine is unstable at higher pH values and ZnO forms at the anode surface).