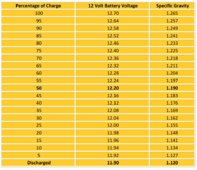
Chemistry does follow the laws of physics- and a lead acid cell (6 of in a '12v' nominal battery in series) gives a known voltage
View attachment 193552
A lead–acid battery's nominal voltage is 2.2 V for each cell. For a single cell, the voltage can range from 1.8 V loaded at full discharge, to 2.12 V in an open circuit at full charge.
In fact there are tables we use (and they haven't changed since I did my apprenticeship 40 years ago) that specifically correlate battery voltages and specific gravity levels with the state of charge... (not that you can measure the specific gravity in many batteries- not without a drill lol)
I REPEAT- I am talking about the rest voltage of a nominal 12v battery when it is fully charged- NOT on charge, not with a load connected, and 'rested' (ie neither charged nor discharged for at least quarter of an hour...)
Charging voltages are a completely different kettle of fish (and can vary with the particular type of battery in use due to the electrolytic carrier in use ie gel/AGM are quite ineffecient as electrolyte carriers and need higher charge voltages than flooded cells), but the chemical reactions involved always produce the same results in voltage output...