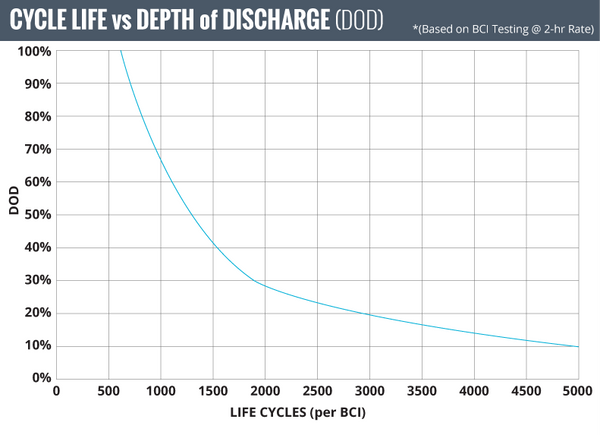
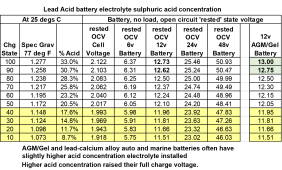
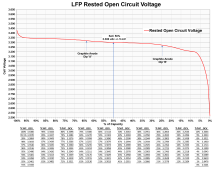
Lead acid battery chemistry is more complex than LFP battery chemistry. This means more chemical conversions, meaning there is more overhead power consumed to drive the chemical conversions causing terminal voltage slump during discharge and voltage bump up during charging.
AGM are a bit better than flooded lead acid, primarily because they have a shorter electrolyte path between plates and slightly higher sulfuric acid electrolyte concentration (about 35% acid at full charge) compared to most flooded lead-acid batteries.
You can only get a determination of state of charge by allowing a battery to reach unloaded rested equilibrium. For lead-acid this takes about 20-30 minutes of rested no load idle period. For LFP cells the time to equilibrium unloaded is less than 3-5 minutes. LFP cells also have a fairly flat electrode voltage, so you need a fairly accurate, good resolution voltmeter with accuracy and resolution to better than a few mVdc. (<0.05% accuracy/resolution)
Both can develop a surface charge that will artificially bump up open circuit terminal voltage and is primarily like a super capacitor after full absorb level charging. This surface charge has very little actual capacity compared to actual battery capacity and burns off very quickly upon some actual load on battery.
Left on float charging for a long period, lead acid batteries will build up a thicker layer of lead-oxide on positive plates that will add some increase in series resistance until discharging burns off some of the thickened lead oxide. You may see an initially greater battery voltage slump when starting discharge that will rise up again before continuing normal discharge voltage drop on lead-acid battery.
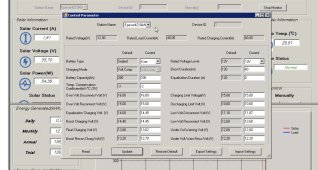
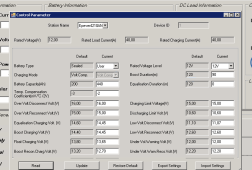
View attachment 148265
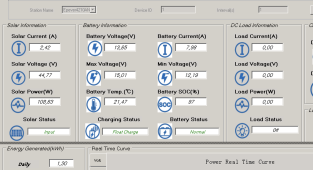
View attachment 148266