Good to know. What about the warnings stating that contact with water would create hydrogen fluoride? That stuff seems super dangerous. What is causing that reaction?
I pulled a couple SDS, and it appears most LFP cells these days are using cells are using Lithium hexafluorophosphate as one of the components of the electrolyte. I can't tell if its by total weight or liquid percentage. Cells can also use LiBOB (Lithium bisoxalatoborate) or LiBF4 (lithium tetrafluoroborate) instead though, so reading the SDS is suggested.
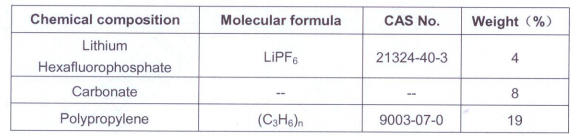
LiPF6 will react with water to produce HF, LiF and phosphoric acid. Hence why you should wash any electrolyte off your skin, and work in a well ventilated area. There may be better solvents for washing skin than water, but I would need to consult a chemist, pure alcohols may work fine.
Atmospheric moisture will react, but its a pretty slow process. The carbonate evaporates readily, shielding the LIPF6. HF gas is much heavier than air.
The real risk is prolonged skin exposure, as water in the skin can react over time and HF can be absorbed. I guess if you took a nap in the room with a bunch of electrolyte that could be a problem.
Some back of the envelope math indicates high toxicity would require direct exposure to more than 1 cells worth of electrolyte.
The stuff is nasty, but its not an brief exposure risk. Use gloves, wash any exposed skin, use good ventilation.
If you have some soaked into the floor or other materials, ventilate the area before entering and during occupation for a week or so.





