autozone, oreillys, best buy etc. recycles batteriesJust in a random dumpster? Lol
I'd just drop it off and run out before they told me "we only do lead acid batteries"
autozone, oreillys, best buy etc. recycles batteriesJust in a random dumpster? Lol
That's Hydrofluoric Acid, hope you didn't get any on you, it readily penetrates skin and can cause severe deep tissue damage and even break down bones. https://www.uthsc.edu/research/safety/documents/sop-hydrofluoric-acid.pdfThe one cell that was drilled all the way through was spilling fluid out.
It was the starting fluid smell I was smelling.
That's Hydrofluoric Acid, hope you didn't get any on you, it readily penetrates skin and can cause severe deep tissue damage and even break down bones. https://www.uthsc.edu/research/safety/documents/sop-hydrofluoric-acid.pdf
1) Skin Contact
HF differs from other protic acids because the fluoride ion readily penetrates the skin, causing
the destruction of deep tissue layers. This process may continue for days if left untreated. Strong
acid concentrations (over 50%), “cause immediate, severe, burning pain and a whitish
discoloration of the skin which usually proceeds to blister formation.” In contrast, the effects of
more dilute solutions may be delayed. The latency period for symptoms (redness, swelling, and
blistering) to appear after exposure to aqueous HF solutions in the 20-50% range may be up to
eight hours. Solutions less than 20% may not produce symptoms for up to twenty-four hours.
Fluoride ions form insoluble salts with calcium and magnesium in bodily tissue. Soluble salts
can form with other cations, which dissociate rapidly causing further disruption and damage to
tissue. The severe, throbbing pain associated with HF burns is thought to result from nerve
irritation due to potassium cations entering the extracellular space to compensate for reduced
calcium ion concentrations.
Fluoride poisoning is associated with hypocalcemia (low calcium levels), hyperkalemia (high
potassium levels), hypomagnesemia (low magnesium levels), and sudden death. Systemic
hypocalcemia should be considered a risk whenever the body surface area of skin burns from
concentrated HF exceed 25 in2 (160 cm2), or about the size of the palm of your hand.
Concentrated HF burns can be fatal if only 2% of the body surface area is exposed.
https://en.wikipedia.org/wiki/Hydrofluoric_acid#Health_and_safety (health and safety section)Sounds like good stuff to add to toothpaste.
I hope I didn't get much on me because I didn't blister up or have redness etc
Someone posted a doc with the composition of the electrolyte in LiFePO4 batteries. Don't recall HF acid being on the list. It was in the post about the battery pack in Germany that was suspected in a house explosion.That's Hydrofluoric Acid, hope you didn't get any on you, it readily penetrates skin and can cause severe deep tissue damage and even break down bones. https://www.uthsc.edu/research/safety/documents/sop-hydrofluoric-acid.pdf
1) Skin Contact
HF differs from other protic acids because the fluoride ion readily penetrates the skin, causing
the destruction of deep tissue layers. This process may continue for days if left untreated. Strong
acid concentrations (over 50%), “cause immediate, severe, burning pain and a whitish
discoloration of the skin which usually proceeds to blister formation.” In contrast, the effects of
more dilute solutions may be delayed. The latency period for symptoms (redness, swelling, and
blistering) to appear after exposure to aqueous HF solutions in the 20-50% range may be up to
eight hours. Solutions less than 20% may not produce symptoms for up to twenty-four hours.
Fluoride ions form insoluble salts with calcium and magnesium in bodily tissue. Soluble salts
can form with other cations, which dissociate rapidly causing further disruption and damage to
tissue. The severe, throbbing pain associated with HF burns is thought to result from nerve
irritation due to potassium cations entering the extracellular space to compensate for reduced
calcium ion concentrations.
Fluoride poisoning is associated with hypocalcemia (low calcium levels), hyperkalemia (high
potassium levels), hypomagnesemia (low magnesium levels), and sudden death. Systemic
hypocalcemia should be considered a risk whenever the body surface area of skin burns from
concentrated HF exceed 25 in2 (160 cm2), or about the size of the palm of your hand.
Concentrated HF burns can be fatal if only 2% of the body surface area is exposed.
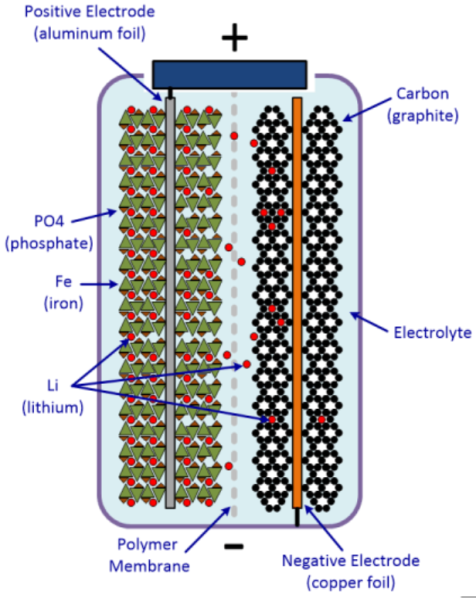
 rebelbatteries.com
rebelbatteries.com
It's formed from the electrolyte reacting with moisture, the carbon coating on the cathode is fluorine doped, when exposed to water the electrolyte releases fluorine gas and hydrofluoric acid. It is a lower concentration (aprox 4-10%) but that can actually be more dangerous, as the are no acute symptoms but it still readily penatrates the skin, and by the time you realize you've been exposed it can already be too late.Someone posted a doc with the composition of the electrolyte in LiFePO4 batteries. Don't recall HF acid being on the list. It was in the post about the battery pack in Germany that was suspected in a house explosion.
Edit: went back to find it. The reference is about Li-ion battery composition, though it under the LiFePO4 section of the website. Another source says this:
"The most commonly used electrolyte is comprised of lithium salt, such as LiPF6 in an organic solution."

How Are Lithium Iron Phosphate Batteries made?
A battery is defined as a device with electrochemical cells to power electrical devices such as lights, mobile phones, and electric vehicles. The positive terminal of a battery is called the cathode, the negative terminal is called an anode. A schematic diagram of battery is shown in Figure 1...rebelbatteries.com
So while fluorine seems to be present, it's part of a salt, not filled with HF.
Here's a SDS for LiFePO4 cells:
isn't hydrofluoric acid the stuff they dissolved bodies in on Breaking Bad?It's formed from the electrolyte reacting with moisture, the carbon coating on the cathode is fluorine doped, when exposed to water the electrolyte releases fluorine gas and hydrofluoric acid. It is a lower concentration (aprox 4-10%) but that can actually be more dangerous, as the are no acute symptoms but it still readily penatrates the skin, and by the time you realize you've been exposed it can already be too late.
Yupisn't hydrofluoric acid the stuff they dissolved bodies in on Breaking Bad?
Yikes!
It's safe as long as it doesn't vent or get drilled into...So you guys are saying it's not safe to use the battery inside my car? ?
I know driving home the other night I had my windows down. I was freezing but rather smell the fresh air vs that battery. It was pretty rough. Can still smell it right now getting in my car but no where near as strong
This is a DIY forum….It's safe as long as it doesn't vent or get drilled into...
Build your enclosure for mobile applications, use separators between the cells, restrain the cells so they don't move and don't charge aggressively and you should be fine.
I wouldn't hesitate to use lifepo4 in a vehicle, I will buy an EV powered by lifepo4 cells when more become available in the states.
Don’t tempt him. Next thing we know he’s gonna have a build thread on how to buy an EV in diy fashionThis is a DIY forum….

I've already spent a few hours looking for motors for an old Sonoma I have as well as a replacement motor for my GTI.This is a DIY forum….

My dream is to have PHEV truck with a huge battery.I've already spent a few hours looking for motors for an old Sonoma I have as well as a replacement motor for my GTI.
The money and time I'd have into swapping the Sonoma wouldn't be worth it. It would hurt to bad to swap the GTI, it's been modifiedjust a little bitheavily...
By the time I get done swapping something I'd be better off buying for a few grand more.
If it leaks out when the cell is punctured, it has not had time to absorb any moisture. So what's leaking out is not HF acid. It might be that over time it reacts with moisture from the air, but not instantaneously.It's formed from the electrolyte reacting with moisture, the carbon coating on the cathode is fluorine doped, when exposed to water the electrolyte releases fluorine gas and hydrofluoric acid. It is a lower concentration (aprox 4-10%) but that can actually be more dangerous, as the are no acute symptoms but it still readily penatrates the skin, and by the time you realize you've been exposed it can already be too late.
yes that's true, it needs some time to react, at which point it becomes hfa and offgasses severely toxic fumes. So like idk, sitting in a hot car truck all day? Do you think that just might qualify?If it leaks out when the cell is punctured, it has not had time to absorb any moisture. So what's leaking out is not HF acid. It might be that over time it reacts with moisture from the air, but not instantaneously.
What settings are you using in the BMS? I've had mixed results with two EEL packs using the (I think) Seplos BMSJust an update here - now that I have had these cells integrated in my system and cycled a few times, I see average battery capacity of around 304Ah for both my packs. I have been charging/ discharging at 175A max using Seplos 200A BMS and all the cells are fairly well balanced using passive balancer alone. Not bad at all for $79 cells!
Having built my first batteries this year...I'd say just go with known good quality cells from a reputable source for your first build. It's quite an involved project as it is, best to try to control for as many variables as possible. Maybe some others more experienced here would say it's fine, just my 2c.I keep flip flopping on these cells for my first build...very tempting. Thinking about order 12 cells, build my first 24v pack and if the other 4 are okay build a 12v pack for a friend.


