Not sure what you mean by saturation, plenty of room for more CO2. And it is not logrithmic.. .its exponential. So every little bit hurts that much more.Reread posts 212, 213.
Cutting Co2 emissions now will not change anything. We are at or near saturation for it's effect. It's logarithmic, not linear incremental.
Only removing Co2 will prevent a new equilibrium, with Miami under water.
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Can Solar & Wind Fix Everything (e.g., Climate Change) with a battery break-through?
- Thread starter svetz
- Start date
Pappion
Retired Engineer Tech
- Joined
- Nov 26, 2020
- Messages
- 559
I think you have the curve backwards. A little has a lot of effect, more has less effect, add more and it tends to flatten out.Not sure what you mean by saturation, plenty of room for more CO2. And it is not logrithmic.. .its exponential. So every little bit hurts that much more.
Effect Saturation point can be debated, is it 95%, 97%, 99%, 99.99%?
svetz
Works in theory! Practice? That's something else
Saturation is one of those words that means different things to different people. For example, CO2 saturation for a plant is the ppm at which more does the plant no additional good (which can be quite high (e.g., 1000 ppm) for some plants, ref).Not sure what you mean by saturation...
But I have seen it used to state that temperature won't go up, which is false... so let's dive into that.
Why temperatures will increase despite CO2 "saturation"
If you go back and look at the saturation graphs in #213 you'll see saturation refers to the wavelengths where the GHG absorbs energy. For CO2, as little as 10 ppm starts absorbing a lot of spectrum whereas the difference between 100 and 1000 ppm is fairly small. So as @Pappion points out, even fairly low levels absorb a lot of heat. So, while technically the spectrum isn't yet "saturated", it's pretty close.So the simple logical conclusion is adding more CO2 can't cause more warming because we're already saturated.
While simple, it's also incorrect. And a good thing too, otherwise we'd all be very crispy.
Back when scientists think production and sequestration were last in balance we were at 280 ppm (also near saturation). Easy to see from the chart in #213 the change between 100 and 1000 ppm is pretty small...so if we were already close to saturation back then... why didn't the world go into catastrophic climate change hundreds of years ago? (e.g., why are we still alive?)
The way I understand it is the atmosphere isn't just absorbing IR Radiation (heat) from the surface. It is also radiating IR Radiation (heat) to space. If these two heat flows are in balance, the atmosphere doesn't warm or cool - it stays the same (that is, the good old 280 ppm days). The trick is that the GHG concentration also affects how much heat can leave the top of the atmosphere. So, as the concentration builds up, the temperature at the lower (surface) layer goes up:
...By adding greenhouse gases, we force the radiation to space to come from higher, colder air, reducing the flow of radiation to space. And there is still a lot of scope for more greenhouse gases to push 'the action' higher and higher, into colder and colder air, restricting the rate of radiation to space even further... ref
Is there a point at which adding more CO2 will not cause further warming?
No. Adding more CO2 to the atmosphere will cause surface temperatures to continue to increase. As the atmospheric concentrations of CO2 increase, the addition of extra CO2 becomes progressively less effective at trapping Earth’s energy, but surface temperature will still rise. ref
The other big negative of increasing the ppm is the very long half-life, what we produce today will be around for a very long time.
Last edited:
You are absolutely wrong on the physics. EVERY increase in ppm of CO2 will increase heat retention in the atmosphere, the physics of gas radiation are pretty clear on that. With pure CO2 you actually become opaque to IR. Now the cumulative effect of that increase is not a linear function of ppm, its ppm to some power. IE exponential. So the increase from 400 to 410 has a larger effect on net temperature than the increase from 390 to 400. Both are increases of 10 ppm, but the delta t will be larger for the 10 ppm at the higher range. Because.... Its an integral problem.Saturation is one of those words that means different things to different people. For example, CO2 saturation for a plant is the ppm at which more does the plant no additional good (which can be quite high (e.g., 1000 ppm) for some plants, ref).
But I have seen it used to state that temperature won't go up, which is false... so let's dive into that.
Why temperatures will increase despite CO2 "saturation"
If you go back and look at the saturation graphs in #213 you'll see saturation refers to the wavelengths where the GHG absorbs energy. For CO2, as little as 10 ppm starts absorbing a lot of spectrum whereas the difference between 100 and 1000 ppm is fairly small. So as @Pappion points out, even fairly low levels absorb a lot of heat. So, while technically the spectrum isn't yet "saturated", it's pretty close.
So the simple logical conclusion is adding more CO2 can't cause more warming because we're already saturated.
While simple, it's also incorrect. And a good thing too, otherwise we'd all be very crispy.
Back when scientists think production and sequestration were last in balance we were at 280 ppm. Easy to see from the chart in #213 the change between 100 and 1000 ppm is pretty small...so if we were already close to saturation back then... why didn't the world go into catastrophic climate change hundreds of years ago? (e.g., why are we still alive?)
The way I understand it is the atmosphere isn't just absorbing IR Radiation (heat) from the surface. It is also radiating IR Radiation (heat) to space. If these two heat flows are in balance, the atmosphere doesn't warm or cool - it stays the same (that is, the good old 280 ppm days). The trick is that the GHG concentration also affects how much heat can leave the top of the atmosphere. So, as the concentration builds up, the temperature at the lower (surface) layer goes up:
The other big negative of increasing the ppm is the very long half-life, what we produce today will be around for a very long time.
As to the atmospheric radiation, you are correct. it radiates back to space. but the re-radiation from the gas is spherically distributed. IE all directions. So not just to space, but back to the ground, horizontally to other bits of the atmosphere.
And yes CO2 is stable in that atmosphere, that is why it took 100s of millions of years to go from a reducing atmosphere to an oxidizing atmosphere. Pre - life, that atmosphere was full of nasty stuff including CO2 and very very little O2.
Pappion
Retired Engineer Tech
- Joined
- Nov 26, 2020
- Messages
- 559
Do you want to save the earth or just the humans?
The Tibetan plateau uplift exposed rock to the atmosphere. The rock absorbed vast quantities of Co2. Sending the earth into a long downward spiral of ice age cycles, which gets progressively worse. Then somehow we learn to dig up the carbon and put it back in the atmosphere and therefore carbon back down to the topsoil. Unwitting Tool or Coincidence?
The Tibetan plateau uplift exposed rock to the atmosphere. The rock absorbed vast quantities of Co2. Sending the earth into a long downward spiral of ice age cycles, which gets progressively worse. Then somehow we learn to dig up the carbon and put it back in the atmosphere and therefore carbon back down to the topsoil. Unwitting Tool or Coincidence?
svetz
Works in theory! Practice? That's something else
Sounds like we're saying the thing, except for the first partYou are absolutely wrong on the physics. EVERY increase in ppm of CO2 will increase heat retention in the atmosphere
IR covers a wide range of the spectrum and CO2 only affects a small portion of the IR wavelength range, see the saturation charts in #213 along with citations. You might also be interested in the spectral ranges of other GHGs covered in #50 & #26.With pure CO2 you actually become opaque to IR.
The IPCC is predicting a 1.5 degree increase at a CO2 level 430 PPM and a 2 degree change at 450 PPM, so with just those two data points, it's easy to see your statement is correct. But, AFAIK, it's for the reasons stated in #229 rather than any correspondingly huge increase in spectral absorption. #229 has cited references, I'd be interested in seeing references to the contrary. Otherwise, we'll just have to agree to disagree.So the increase from 400 to 410 has a larger effect on net temperature than the increase from 390 to 400.
Yup. But, it's only the heat emitted to space that leaves the system (e.g., provides "cooling")....but the re-radiation from the gas is spherically distributed. IE all directions. So not just to space, but back to the ground, horizontally to other bits of the atmosphere.
Personally, I don't think either is in jeopardy unless there's some secondary effect (e.g., triggers a nuclear war (and even then both would probably survive)). I'm up for holding the line on costs. Flood insurance in Florida is already ridiculously costly. But if big coastal cities around the world (or states like Florida) have to have the population relocated, if small wars get fought due to populations escaping unlivable conditions or starvation.... well .... the costs would be monumental. So, a war-time-like burden for a couple of decades to prevent far larger future costs for centuries to come... yeah... I'd be happy to pay my fair share.Do you want to save the earth or just the humans?
Mainly though, I don't worry about it all. It's just something interesting to think about and try to understand the science. Unless someone comes out with rejuv treatments, none of this is going to affect me. Technology seems like it is ever accelerating. People are aware of the problems and they're making things happen.
svetz
Works in theory! Practice? That's something else
Great question! I started a thread on it because... I don't know.[if]... climate change is coming ... What are our options ? my vote at the poll is just one tiny voice.
What Can I personally do to help against climate change?
svetz
Works in theory! Practice? That's something else
I don't think anyone can claim with certainty a net reduction in carbon emissions.In reality fracking greatly increased the supply of natural gas where the reduced cost replaced coal in power plants. This resulted in a net reduction of carbon emissions.
Apologies for responding in a different thread, but didn't want to derail the other one with climate change mechanisms as it would be off-topic there.
Coal emits about twice the lbs of CO2 per BTU as natural gas (ref), so it's is pretty easy to see the carbon impact by switching fuels.
Basically natural gas gets a "boost" as the hydrogen in it is also converted which gives off heat. So I believe Delmar is correct about it having a net reduction of carbon emissions for fuel types. From the graphs carbon emissions never went down or slowed, but that's probably because we made more energy and the transition took a while.
Also worth noting is burning coal also emits more SO2 which is a very powerful anti-greenhouse gas (#15). SO2 is also responsible for acid rain which is thought to harm forests (a carbon sequestration mechanism). On top of that, burning natural gas also emits a lot of water into the atmosphere with that hydrogen where it can act as an anti-greenhouse gas or greenhouse gas (#20).
So, while the carbon numbers are easy to see for the two fossil fuels, the actual climate impacts are a lot more convoluted. The good news is, the IPCC climate models try to take those into consideration and have been very accurate (#32).
wattmatters
Solar Wizard
No prob. I don't propose to come back here though so this will be it.Apologies for responding in a different thread
I was not talking about relative CO2 emissions from the combustion processes, that's just basic chemistry and is not in doubt.Coal emits about twice the lbs of CO2 per BTU as natural gas (ref), so it's is pretty easy to see the carbon impact by switching fuels.
I was referring to fugitive emissions from the extraction of coal seam gas, which are substantial and it's a terribly potent GHG (20 times worse than CO2). When you factor this in then it evens up the GHG scales considerably, so much that it's not actually clear cut which is worse from a greenhouse perspective. The problem is we don't actually measure fugitive emissions very well, and it's likely to be significantly worse than the fracking companies claim (fossil fuel companies are not exactly known for claiming the pollution they cause is worse than it actually is).
I take your point about other pollutants from use of coal, that I do not dispute (and living in a state which is a big user of coal power we are all too aware of the pollutants). But replacement with gas is not the solution. Central to my main point is we should be leaving as much of both in the ground as we can.
svetz
Works in theory! Practice? That's something else
The population chart in the IPCC (shown right) covers a wide range. From a scary (19 billion by 2100 in the worst-case scenario to a population decline in the best-case scenario. As far as I can tell it's based on current population/urbanization trends tempered by death rates from diseases and medical improvements. The IPCC scenarios represent various guesses from what's the best we could possibly manage to do; to what if we do nothing. A lot of what's in the new report are descriptions as to what will happen based on those models. | 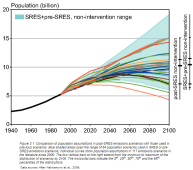 |
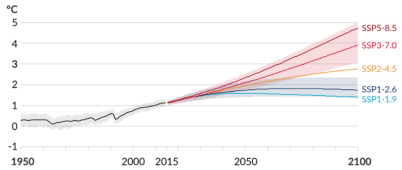
The scenarios for temperature look like this:
Each scenario has a population curve behind it that you can see from the scenarios link. Most cases predict a population peak around 2060 and then a decrease (the best case predicts a peak around 2045). In addition to the population curve are other factors, such as how much progress the world has made towards net-zero.
ROFL. I've read the short one for policy makers front to back to get the high level and spelunked into the full report to dig up things I had questions on....thank you for reading the huge report....
Hedges
I See Electromagnetic Fields!
- Joined
- Mar 28, 2020
- Messages
- 21,607
Fortunately, there are people working behind the scenes to save the world for the rest of us.

 news.yahoo.com
news.yahoo.com

Photos show the lineup of private jets that the world's elite, including Jeff Bezos, took to the UN's climate conference in Scotland
Private jet travel remains a blind spot for world leaders and the wealthy when discussing climate change.
Fortunately, there are people working behind the scenes to save the world for the rest of us.

Photos show the lineup of private jets that the world's elite, including Jeff Bezos, took to the UN's climate conference in Scotland
Private jet travel remains a blind spot for world leaders and the wealthy when discussing climate change.news.yahoo.com
But they are impotent, so they are allowed to pollute more because they are helping more. Or something like that.
svetz
Works in theory! Practice? That's something else
But they are impotent, so they are allowed to pollute more because they are helping more. Or something like that.
Something like that.
The problem with hatchet entertainment-news jobs like that is there's no real investigation as to if any of them, like Bezos, have carbon offsets to make their air travel carbon neutral. If they're personally paying to have their jet's carbon footprint removed or using green fuels then that's far better than your average family flying somewhere for vacation.
Also, at COP26, Bezos pledged $2B. That's in addition to the $10 Billion in the Earth Fund. It's more than I've committed so I won't begrudge him an airplane ride. He went there and ponied up.
Rather than poke fun at private citizens actively spending their own $ to make things better, why not turn to the BBC leak for climate change amusement? (BTW, if you read it don't freak out ... for example I'm sure references to "Austrailia" refer to the federal government which is not on board with the program whereas every state and territory is, probably some changes coming in the upcoming elections. Norway's complaint about CCS confused me (thought it was part of the plan for 1.5), now I have to go check ; -).
CCS Update: If I'm understanding it right, the IPCC report does say we need CO2 removal to meet the 1.5C target, but they leave the "how" open-ended as Carbon Dioxide Removal (CDR) technologies whereas CSS refers to a subset of technological approaches to Carbon Capture Storage solutions. There's a doubt on those as they take energy and some claim they only distract from the real solutions. Possibly some of the controversy is from recent discoveries such as @robby's video post where planting trees is worse in the short term.
Last edited:
wattmatters
Solar Wizard
Don't hold your breath for a change of government, nor for a dramatic shift in policy if there is a change.BTW, if you read it don't freak out ... for example I'm sure references to "Austrailia" refer to the federal government which is not on board with the program whereas every state and territory is, probably some changes coming in the upcoming elections.
Just to give you some idea of how idiotic the Australian presence at COP26 is, the official Australian booth is sponsored by one of Australia's biggest oil and gas companies, Santos.
We will see how China handles this, as there seems to have a lot of will, from top-down authority as well as from common people, and certainly the know how and resources. China's biggest hurdle/challenge is the massive population, every mouth wants to be fed and every body likes comfort, and deservedly so. Hopefully our wasteful lifestyle import can be tempered. Solar & wind plus storage can support a certain populous at a certain lifestyle. Homo sapiens are just out of balance and Mother is spanking us
Sort of sounds likeSomething like that.
The problem with hatchet entertainment-news jobs like that is there's no real investigation as to if any of them, like Bezos, have carbon offsets to make their air travel carbon neutral. If they're personally paying to have their jet's carbon footprint removed or using green fuels then that's far better than your average family flying somewhere for vacation.
Also, at COP26, Bezos pledged $2B. That's in addition to the $10 Billion in the Earth Fund. It's more than I've committed so I won't begrudge him an airplane ride. He went there and ponied up.

Indulgence - Wikipedia
Hedges
I See Electromagnetic Fields!
- Joined
- Mar 28, 2020
- Messages
- 21,607
The problem with hatchet entertainment-news jobs like that is there's no real investigation as to if any of them, like Bezos, have carbon offsets to make their air travel carbon neutral. If they're personally paying to have their jet's carbon footprint removed or using green fuels then that's far better than your average family flying somewhere for vacation.
It's like governor of California or mayor of Oakland issuing a mandate to wear masks indoors regardless of vaccination status.
Then immediately going to a party maskless.
And declaring, "It's OK, everyone here is vaccinated."
Remember the time U.S. automobile CEOs flew by private jet to Washington with their hands out for a federal bailout?
They were told leave, come back by car.
The example these obscenely rich guys show is worth more than any agreement made, such as 1st world doing without and paying money while (formerly) 3rd world expands coal-powered generation.
The rest of us have been meeting by Zoom.
The climate conference could have been held by Zoom, except for those who could attend in person traveling by electric rail.
"Carbon Offsets"
Meaning what, he bought power generation credits from the PV panels I have in my yard?
He paid to have trees planted somewhere (after the bookkeeping was complete, they were cut down by locals)?
As it said, (partially) renewable fuels. Meaning palm oil, from a plantation grown where rainforests were burned down to get the agricultural land?
To the extent these guys built companies providing home delivery of products (with less emissions that you driving to the mall), making cars that run on renewable energy, computers and data centers which are more efficient, that is their contribution.
svetz
Works in theory! Practice? That's something else
Loved this video from @Supervstech, thought they did a good job on it.
One of the things that struck me as odd in the COP26 stuff I've read is the lack of anything decrying Climate Change as hoax. There was plenty of arguing and disagreement that made me think the 1.5C line wouldn't hold, but not because people weren't believing. Pity Russia & China didn't make it.
One of the things that struck me as odd in the COP26 stuff I've read is the lack of anything decrying Climate Change as hoax. There was plenty of arguing and disagreement that made me think the 1.5C line wouldn't hold, but not because people weren't believing. Pity Russia & China didn't make it.
Green fuels are sustainable, so it seems like a reasonable way to go about it to me. But, to each their own.[green fuels/carbon offsets] Sort of sounds like indulgence...
I differ in opinion on that. One is sustainable and carbon neutral. The other is hypocrisy. Sure, takes more money to be green on your private jet, but takes money to have a private jet too. No crime in being rich and I'd only think them hypocritical if they weren't green in the process.[using green fuels] ... It's like governor ...issuing a mandate to wear masks ... Then immediately going to a party maskless.
Last edited:
Hedges
I See Electromagnetic Fields!
- Joined
- Mar 28, 2020
- Messages
- 21,607
The fuel they used in their jets to fly to a conference about climate change wasn't sustainable, green fuel. It was a blend, partly good old fossil fuel.
I don't think any aircraft are run 100% on sustainable fuel.

 skynrg.com
skynrg.com
It can be blended up to 50%
 www.bp.com
www.bp.com
So they're only being half as wasteful.
Betcha there isn't enough sustainable fuel to blend 50% with all jet fuel. They used some, reducing what could be used in other, fully loaded flights.
If so, all told, their impact on fossil fuel consumption and CO2 emission was identical to if they flew their private jets with 100% fossil fuel.
I don't think any aircraft are run 100% on sustainable fuel.

Sustainable Aviation Fuel | SAF | SkyNRG
Sustainable aviation fuel is made from sustainable resources that can be mixed with fossil jet fuel to reduce emissions. Learn more about SAF.
It can be blended up to 50%

What is sustainable aviation fuel (SAF) and why is it important? | News and views | Air bp
Sustainable aviation fuel – what is it and why is it important? Tom Parsons, Air BP’s Biojet commercial development manager, explains what sustainable aviation fuel is (SAF) and gives his view on whether SAF really is the answer to reducing aviation emissions.
So they're only being half as wasteful.
Betcha there isn't enough sustainable fuel to blend 50% with all jet fuel. They used some, reducing what could be used in other, fully loaded flights.
If so, all told, their impact on fossil fuel consumption and CO2 emission was identical to if they flew their private jets with 100% fossil fuel.
svetz
Works in theory! Practice? That's something else
| Was looking for some information on Methane and found the graphic to the right in a CSR. Interesting that while CO2 is going up, we've actually reduced CH4 by nearly 10%. Most of that has been from landfills and tightening up the fossil fuel sectors. 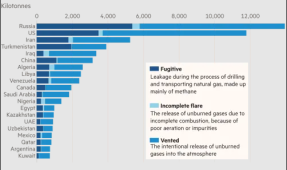 The amount of "fugitive" vs. "vented" is interesting. Seems like the vented is something we should be able to get a handle on. I did see something regarding biosolids (microbes in the dirt) that can eat the methane and convert it into CO2 before it escapes a landfill. | 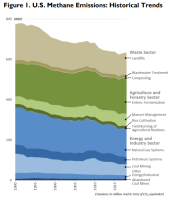 |
svetz
Works in theory! Practice? That's something else
Couldn't respond in the other thread as it would be off-topic.While methane is a potent greenhouse gas, it is also destroyed quickly in the atmosphere through chemical reactions. Carbon Dioxide is, more or less, a permanent problem, but methane only lasts for around a decade before being broken down by natural processes.
But, from post #9 methane is a bigger contributor than CO2 by 375% at its 2019 concentrations. Methane has a half-life of 9.1 years, so it's not like everything we release today is gone within a decade (e.g., the 800 MMT in 1990 accounted for 200 MMT 20 years later). Unfortunately, it breaks down into CO2 which can be sequestered by natural processes (although the half-life is very long).
Half-life is relatively unimportant when you're putting in more than comes out. E.g., you put 10 drops in and one leaks out... you still have a buildup of 9 drops. So, as long as we're emitting it'll be adding to the surface temperature and we'll always be emitting some. From the graphs above, looks like some of that (~100 MMT) is low-hanging fruit.
svetz
Works in theory! Practice? That's something else
Do you have a better link? Never seen anything about that (there are aticles that methane kills grass though).... grass pulls the methane from the air, Joel Salatin...
That link lists an author, but i couldn't find anything about grass pulling methane from the air.
Everything I see (ref) is the usual yada yada:
BTW, the same reference does say managed grasslands are better (even if the overall net is positive).Grasslands absorb and release carbon dioxide (CO2), emit methane (CH4) from grazing livestock, and emit nitrous oxide (N2O) from soils.
thanks for posting this, didn't appreciate that non-Energy sector has a sizable contribution. CH4 mitigation does seem like a low hanging fruit. Is livestock methane accounted for as Enteric Fermentation ?
Was looking for some information on Methane and found the graphic
to the right in a CSR.
View attachment 71621
svetz
Works in theory! Practice? That's something else
I was surprised too! I can see why they're going after it. Ran across this satellite methane map which was sort of surprising. What surprised me is some states (e.g., North Dakota) have no-venting laws, CA reports very low emissions (but looks pretty high), Colorado is beautifully divided by the mountain range, etc. | 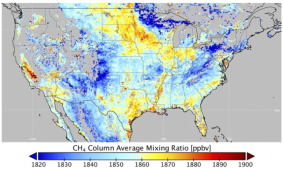 |
Technically it's any anaerobic fermentation by bacteria, but that chart splits out some, such as rice, landfills, composting, and wastewater treatment.Is livestock methane accounted for as Enteric Fermentation ?
So, as I read it that 200 MMT from last year is primarily burps and farts from livestock.
Another benefit of fake meat is that less livestock means less livestock food production which means less fertilizer, which means less N2O (which is even a more powerful GHG than CH4 and has a century-long half-life).
Similar threads
- Replies
- 169
- Views
- 5K
- Replies
- 10
- Views
- 577
- Replies
- 13
- Views
- 717
- Replies
- 5
- Views
- 717