danielfp248
Battery researcher
- Joined
- Sep 7, 2020
- Messages
- 429
I'm gonna address a few things in this one post, its simpler that way.
1)
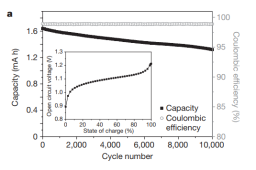
Point being, if we consider the battery's above, then the MnFe has to be able to compete and its here that, at least I, is fumbling in the dark. I have no cyclability reference besides little to no degradation at 500 cycles. So before one worry about the cost for hundreds of kg, I would want the base knowledge to be known
- If we look at a 48V@10kWh Li-ion battery pack from LG Chem, then 7500€ is a fair price. Here we can expect roughly 3-5k cycles.
- If we then look at similar LFP battery pack, then we land between 5500-7500€ but get substantially longer cycle life. LFP's are estimated to return 5000-10,000 cycles.
- A 10kWh NiFe pack on the other hand will cost roughly 10,000€, but there, >10,000 cycles is no issue.
- An all iron salt battery, the FeCl, is estimated to do more than 20,000 as in little to no losses are seen at those cycling's - but I currently do not have a cost projection here. So lets use NiFe levels.
2). Stumbled upon a paper where they used an anion-exchange membrane in a FeCl battery at pH of 2-3 (Tokuyama A901, 11 μm thickness) which was stable for at least 50 cycles.
Ttitle: ... A High Efficiency Iron-Chloride Redox Flow Battery for Large-Scale Energy Storage - DOI 10.1149/2.0161601jes ... curious about your thoughts here.
and here is a PDF with reference to the membrane: https://www1.eere.energy.gov/hydrogenandfuelcells/pdfs/amfc_050811_fukuta.pdf
also some info here http://www.astom-corp.jp/en/product/02.html
ASTOM & Tokuyama merger information: http://www.membrane-guide.com/membrane_separation/ion-exchange/japan_ion_exchange_membrane.htm
3) The colour shifts sounds mesmerising
An important point is that both NiFe and FeCl3 flow batteries are very maintenance heavy.
I don't know if you have used NiFe batteries before, but periodic electrolyte changes - which involves moving and dumping large amounts of highly concentrated KOH+LiOH solutions - need to be done, often several times per year. This is especially true for some of the modern chinese versions of NiFe batteries, which are constructed much more cheaply than those that Edison made early on. While NiFe might sound great in some aspects, in practice it is a quite annoying battery chemistry to run and maintain. I have run these batteries before and I would say they are anything but care-free.
In the case of FeCl3 flow batteries, a similar problem happens. Due to the H2 evolution at the anode, you will need to constantly adjust the pH of the anolyte so that you don't start seeing hydroxide precipitation issues. These pH adjustments would need to be done practically every 50 or so cycles of the battery. Thankfully this process is much more prone to automation, given that you just need to keep the pH at a low value.
Using an anion exchange membrane would be ideal, but considerably more expensive than the commonly used cation exchange membrane types. Conductivity of anions is also slower than protons, so this means you also get a penalty in terms of current density.
Another issue is that since you're plating a metal, your capacity does not scale solely with the volume of electrolyte but you also need to increase the physical volume of the anodes to get more capacity. This imo, is counter productive since the ideal of a flow battery is to fully decouple power density and energy density of the setup.