If we select a refrigerant which condenses well below Minnesota winter temperatures, can we get good heat-pump efficiency?
CH4? O2? N2? Something else?
The existing R410a would work.
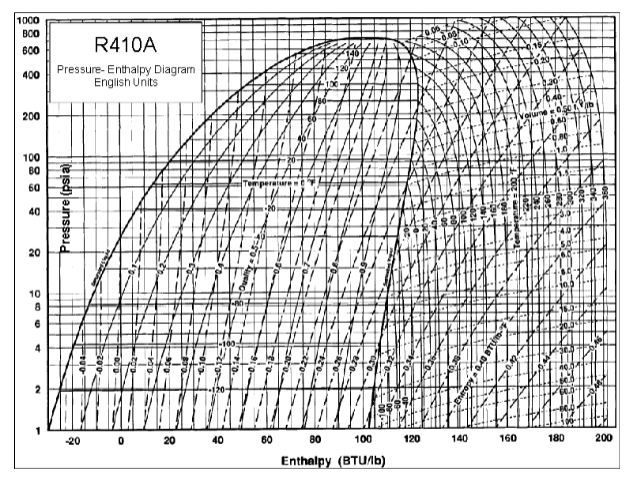
The evaporator pressure for a "standard" heat pump is probably set to around ~100 psia, so from the R410A pressure enthalpy chart below, that's a fixed temperature above freezing, and probably set that way so the outside coils don't freeze up (for those units that don't have defrost cycles as
@MurphyGuy said). Probably around 90 psia for those that do (a boiling temperature of 20F), If the outside temperature is close to or lower than the boiling temperature obviously no heat can flow into the system (that is if the evaporator boils at 36 degrees and it's 30 degrees outside, heat flows from hot to cold, so heat couldn't flow into the evaporator to do its job).
| Svetz's two-second guide to PE charts
The chart to the left represents the pressure, temperature,
and enthalpy of a gas. Given any pressure you can find
the boiling temperature and calculate the change in
enthalpy (heat) at that pressure between gas and liquid
phases.
The "hump" is the interesting part. The left side is a
fluid (e.g., water) and the right it is a gas (e.g., steam).
Because the transition between phases at constant pressure
is a constant temperature, the horizontal lines across the
hump are the temperature.
So, at 90 psia, the phase change is about 120-20, or 100
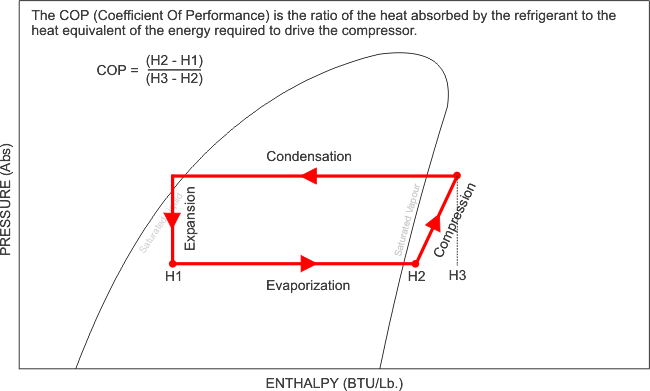
BTU/lb. The red line in the diagram below shows a full cycle.
|
As you can also see from the chart above, if you drop the evaporator pressure to 25 psia, then the evaporation temperature is -40F, so it'll work in a lot colder environment.
Different refrigerants have very different properties. For example, water to water is 970 BTU/lb at 1 atmosphere (
PE Chart). I believe water is the "king" of heat moved per lb. Also non-toxic!
So why don't they all go to 25 psia for -40F? To get to the condensation temperature (where the heat comes out) you always need to compress it to around 310 psia to get ~100F. The larger the pressure delta, the more work the compressor needs to do. So it will become slightly less efficient.
If the expansion valve were dynamic to where you could set evaporator pressure, then the system could dial in the optimum pressure for the outside temperature and ambient humidity (e.g., defrost cycles).
In Summary
Theoretically, heat pumps can work at any temperature. Practical concerns (like coils icing up) prevent them from working as efficiently with air as they need defrost cycles. But, getting heat from other mediums such as being buried solves that problem. Going deeper for geothermal heat rather than ambient heat makes them even more efficient.