Personally, I won't charge below 2 degrees celsius. If it's that cold, how much sunshine is actually available? I'd rather stay on the safe side. But yeah, it sure is interesting to imagine pushing the limits of this chemistry. So cool.
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
How to Parallel Balancing. (YEP 99% of us is doing it wrong)(PART#1)
- Thread starter ghostwriter66
- Start date
ghostwriter66
"Here - Hold my Beer"
But...would you?
NOPE -- and the magic question I am going to ask the DELIGREN engineers the moment they get back from Chinese New Year is "what special "thing" needs to be done to charge below 32F".. if they say NOTHING then i will apologize to everyone ... If they say you need to charge at 0.001 amps for 100 hours - then someone owes me a beer...
Wait guys. What if you have a active balancer on your system... And your BMS cuts off charging, but as you discharge, and your pack goes out of balance at low SOC, and triggers active balancer, and starts charging a individual cell! At an unsafe temperature. Haha I never thought of that before! Well, if it's discharging quickly, the internal cell temperature would probably rise enough to enable charging.
SolarRat
Solar Addict
.. if they say NOTHING then i will apologize to everyone ...
If they say "nothing", I personally would still be skeptical. Not that I don't think it's possible, but because I don't think they (the customer support team) know or care.
SolarRat
Solar Addict
Since we are already way off topic, here is some good research data on charging near or way below freezing:

 www.pnas.org
www.pnas.org
" Schimpe et al. (11) cycled identical graphite/LiFePO4 cells at different temperatures. Cells at 25 °C lost 8% capacity in 2,800 equivalent full cycles (EFCs). At the same capacity loss, cell life drops to 1,800 EFCs at 15 °C, 1,400 EFCs at 10 °C, and 350 EFCs at 0 °C. "
Still want to try -5c?
There are literally thousands of these research papers out there for us to gather info from, a bit here and a bit there....

Fast charging of lithium-ion batteries at all temperatures
Range anxiety is a key reason that consumers are reluctant to embrace electric vehicles (EVs). To be truly competitive with gasoline vehicles, EVs should allow drivers to recharge quickly anywhere in any weather, like refueling gasoline cars. However, none of today’s EVs allow fast charging in...
" Schimpe et al. (11) cycled identical graphite/LiFePO4 cells at different temperatures. Cells at 25 °C lost 8% capacity in 2,800 equivalent full cycles (EFCs). At the same capacity loss, cell life drops to 1,800 EFCs at 15 °C, 1,400 EFCs at 10 °C, and 350 EFCs at 0 °C. "
Still want to try -5c?
There are literally thousands of these research papers out there for us to gather info from, a bit here and a bit there....
Last edited:
Max Bender
New Member
- Joined
- Dec 21, 2019
- Messages
- 78
WOW, thank you. I'm looking forward to Pt.2So my boss who has a PhD in Chemical Engineering went to China on business about a month ago and visited one of the largest LiFePO4 battery manufacturers in the world that we use. They gave him a very good tour and today we reciprocated their hospitality by hosting their Lead Design Engineer who is in the States for an upcoming Solar conference. It was a VERY interesting day with him with lots of hands on "Show and Tell". So I thought I would pass what I can on to those interested.
MOST OF THIS INFORMATION IS TAKEN/COPIED FROM THE SLIDE DECKS FROM HIS CLASS
How to Parallel Balancing. (YEP 99% of us is doing it wrong)
So we have all heard the instruction that by just wiring the cells in parallel they will somehow all get themselves magically in balance. This is NOT entirely true unless you have several days-weeks to waste. Now let me caveat something first. 95% of all the LiFePO4 (LFP) cells coming out of China now are made in batches and shipped in batches. This means that if you order four 100aH batteries that the odds are they are going to all be charged at 3.2v at the factory and by the time you get it ALL the cells are probably within a 0.1V variance. SO simply paralleling those four batteries for the next 24 hours will probably do the trick.
BUT if you get batteries that are 0.25v or more out of whack - or you don't want to wait 24 hours - here's how the Manufacturing Design engineers recommend. Remember - Balancing requires a voltage differential to move current between or from/to the cells. That’s why just putting them together in parallel and leaving them does NOT do much.
So to QUICKLY achieve a PERFECT balance.
1. If possible - top each of the cells up to the voltage of the highest cell prior to wiring them in parallel.
2. Wire the cells in parallel
3. Set the power supply to 3.40V and the amperage to about 80% of the max it can do. (Setting it at 80% is only to prolong the life of the Power Supply)
4. Turn on power supply and charge to 3.40V
5. When the current has dropped to 0.0A at 3.40V turn off the power supply & set it to 3.50V
6. Turn on power supply and charge cells to 3.50V
7. When current has dropped to 0.0A at 3.50V turn off the power supply & set to 3.60V
8. Allow current to drop to 0.0A at 3.60V
9. Disconnect Power.
10. The Battery pack is now perfectly balanced.
NOTE: Due to LFP characteristics, It will take about 70% of the time to get from 3.2 to 3.4V. Then about 25% of the time to get 3.4 to 3.5V... then 3.5 to 3.6 will be very short ...
NOTE: Measure the input voltage right at the cells - do not trust the voltmeter on the power supply.
NOTE: You have to do each step as described – you CAN'T simply set it at 3.6V and walk away.
I am sure this will work, but if testing shows stepping up the rate is faster, and better for the battery, why not?I agree that we need to balance at high SOC, but I don't understand the purpose of doing it in steps. They can handle the charge rate just fine. Setting power supply to 3.65v and waiting for current to drop will top balance all of the cells.
Why would it be faster? It should be slower due to ohms law. Higher voltage means faster power transfer. And it's well within c rate of cell.I am sure this will work, but if testing shows stepping up the rate is faster, and better for the battery, why not?
FilterGuy
Solar Engineering Consultant - EG4 and Consumers
Because they were NOT balanced when you made them parallel
I could believe current flowing, but a voltage difference?
Unless there is resistance between the cells and current flowing, I don't see how there would be a voltage difference between the cells.
If there is a measurable difference either the resistance or the current (Or both) would have to be pretty dang significant.
Solarfun4jim
Solar seduced :-)
Maybe my understanding of 'active' balancers is wrong, but i thought that they 'actively' balanced all the time on both charging and discharging, ensuring that the cells stayed balanced all the time. I didnt think it was something that was 'triggered' at set points etc. So i'm thinking, the pack never goes out of balance in the first place.... but i'm only just trying to get to grips with this stuff(no experience).Wait guys. What if you have a active balancer on your system... And your BMS cuts off charging, but as you discharge, and your pack goes out of balance at low SOC, and triggers active balancer, and starts charging a individual cell! At an unsafe temperature. Haha I never thought of that before! Well, if it's discharging quickly, the internal cell temperature would probably rise enough to enable charging.
With passive balancers, that gets done at the top to passively bring them into line, but not with active....(i think)
I know i would like a fuller explanation Will, if i'm totally wrong on this.
FilterGuy
Solar Engineering Consultant - EG4 and Consumers
So what is unknown? If the spec calls something out why should you not believe it? What other specs should I not believe? Max voltage? Min Voltage? Or do we get to selectively choose which ones to believe or not believe?And that's the way we should all treat these unknowns. Better safe than sorry until there are definitive answers.
If I saw one spec that was out of line with a everyone else, I think there is room for concern. However, I have seen a LOT of cells speced at -5. Just because that does not fit someones belief system does not mean it is wrong.
Would I charge at -5C, No. I would probably set my BMS to cut off before that, just like being conservative with any other spec.
Solarfun4jim
Solar seduced :-)
Yeah, but they dont state implicitly that you can charge down to that temp at 1C, just that you can charge down to that temp. They are not stating any falsehoods, maybe just economical with all the facts perhaps???So what is unknown? If the spec calls something out why should you not believe it? What other specs should I not believe? Max voltage? Min Voltage? Or do we get to selectively choose which ones to believe or not believe?
If I saw one spec that was out of line with a everyone else, I think there is room for concern. However, I have seen a LOT of cells speced at -5. Just because that does not fit someones belief system does not mean it is wrong.
Would I charge at -5C, No. I would probably set my BMS to cut off before that, just like being conservative with any other spec.
FilterGuy
Solar Engineering Consultant - EG4 and Consumers
Another interesting note he said was that really only the American's seem obsessed with cell balancing. He said that he only uses BMS for High Current cutoff, Low Current Cutoff, and Low/High Temperature cutoff -- he said that if you get 4 batteries from the same batch that them getting enough out of "whack" (my word) to be concerned about would take a long time ...
This is something I have come to believe as well... If you you have reasonably matched cells, why do you need to top or bottom balance at all? As far as I can tell, balancing really only allows you to squeeze those last few electrons in at the top of the charge and get those last few electrons out at the bottom of the discharge. If your voltages are set reasonably conservative, What difference does it make? Also, if you are using a system with a balancing BMS, it is going to redo it all over time anyway.
If you are powering an RC plane, I can see you would want to balance.... this is where people are going to try to squeeze as much as they can out of the battery. That is not what most of us Willites are doing. Our priorities are almost the opposite. Rather than squeeze every bit out, we want to make the batteries last, so we are going to be setting the voltages conservative.
Steve_S
Offgrid Cabineer, N.E. Ontario, Canada
Excelent Thread and good discussion.
Just to toss in a bit of randomness and/or monkey wrench, just for kicks & giggles.
What if you have a BMS which is capable of balancing cells During Charging / Discharging and while in Storage (no amps being drawn) States ? Example BMS is the Chargery which does so if enabled. It isn't a big active balancer, being only 1.2A but seemingly capable for most use cases I would think. Seems to me, if you need more powerful balancing than that, there are likely some issues with the cells.
Link for the Progressive Dynamic products for those who may be interested:
https://www.progressivedyn.com/specialty/pd9100l-series-lithium-converters/
Just to toss in a bit of randomness and/or monkey wrench, just for kicks & giggles.
What if you have a BMS which is capable of balancing cells During Charging / Discharging and while in Storage (no amps being drawn) States ? Example BMS is the Chargery which does so if enabled. It isn't a big active balancer, being only 1.2A but seemingly capable for most use cases I would think. Seems to me, if you need more powerful balancing than that, there are likely some issues with the cells.
Link for the Progressive Dynamic products for those who may be interested:
https://www.progressivedyn.com/specialty/pd9100l-series-lithium-converters/
The PNAS citation is very interesting. I had previously thought that 0 C charging would result in an immediate "catastrophic" capacity loss. This suggests 8 percent loss after 350 cycles at freezing. I will need to read the paper and see how evenly the accelerated low temperature loss is spread across those 350 charge cycles. If it is not front loaded too much, the consequences of one or two cold days might not be as grim as I had thought (although obviously to be avoided if possible).Since we are already way off topic, here is some good research data on charging near or way below freezing:

Fast charging of lithium-ion batteries at all temperatures
Range anxiety is a key reason that consumers are reluctant to embrace electric vehicles (EVs). To be truly competitive with gasoline vehicles, EVs should allow drivers to recharge quickly anywhere in any weather, like refueling gasoline cars. However, none of today’s EVs allow fast charging in...www.pnas.org
" Schimpe et al. (11) cycled identical graphite/LiFePO4 cells at different temperatures. Cells at 25 °C lost 8% capacity in 2,800 equivalent full cycles (EFCs). At the same capacity loss, cell life drops to 1,800 EFCs at 15 °C, 1,400 EFCs at 10 °C, and 350 EFCs at 0 °C. "
Still want to try -5c?
There are literally thousands of these research papers out there for us to gather info from, a bit here and a bit there....
KG6S
ELI the ICE man
What are Y’all using for a power supply?
Excelent Thread and good discussion.
Just to toss in a bit of randomness and/or monkey wrench, just for kicks & giggles.
What if you have a BMS which is capable of balancing cells During Charging / Discharging and while in Storage (no amps being drawn) States ? Example BMS is the Chargery which does so if enabled. It isn't a big active balancer, being only 1.2A but seemingly capable for most use cases I would think. Seems to me, if you need more powerful balancing than that, there are likely some issues with the cells.
Link for the Progressive Dynamic products for those who may be interested:
https://www.progressivedyn.com/specialty/pd9100l-series-lithium-converters/
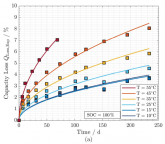
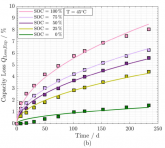
Here is an interesting graph from the Schimpe paper showing 5% loss of capacity when storing LFP 100% SOC at 35 C for ~200 days, and a second showing how progressively lower SOC storage affects this.Since we are already way off topic, here is some good research data on charging near or way below freezing:

Fast charging of lithium-ion batteries at all temperatures
Range anxiety is a key reason that consumers are reluctant to embrace electric vehicles (EVs). To be truly competitive with gasoline vehicles, EVs should allow drivers to recharge quickly anywhere in any weather, like refueling gasoline cars. However, none of today’s EVs allow fast charging in...www.pnas.org
" Schimpe et al. (11) cycled identical graphite/LiFePO4 cells at different temperatures. Cells at 25 °C lost 8% capacity in 2,800 equivalent full cycles (EFCs). At the same capacity loss, cell life drops to 1,800 EFCs at 15 °C, 1,400 EFCs at 10 °C, and 350 EFCs at 0 °C. "
Still want to try -5c?
There are literally thousands of these research papers out there for us to gather info from, a bit here and a bit there....
Xiryx
New Member
- Joined
- Nov 23, 2019
- Messages
- 27
What are Y’all using for a power supply?
I used a Mean Well HRP-300-3.3 for top balancing 3.2 V cells in parallel. Worked perfectly for me.
[Edit] One issue I had using the Mean Well was the lack of metering. A nice benchtop power supply offers the advantage of readouts, but I couldn't justify the $$$ for this use. I also realized my AH meter wouldn't operate on less than 5V and didn't support using an external power supply. Fortunately I was able to measure amps using my shunt, a volt meter, and good old Ohm's law. This will do the job for my once a year top balance. If I were doing it more often, I'd certainly pick up a cheap watt meter that can either run on 3V or off an external power source.
Last edited:
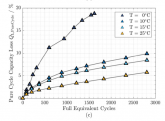
Here is the relevant figure from the Schimpe paper - it seems that the loss of capacity when charging at 0 degrees C is quite rapid, but one ore two charges at freezing would not "kill" the battery. Sadly no data in this paper on charging below freezing. Of note, this test was done at a high 1C charge rate.The PNAS citation is very interesting. I had previously thought that 0 C charging would result in an immediate "catastrophic" capacity loss. This suggests 8 percent loss after 350 cycles at freezing. I will need to read the paper and see how evenly the accelerated low temperature loss is spread across those 350 charge cycles. If it is not front loaded too much, the consequences of one or two cold days might not be as grim as I had thought (although obviously to be avoided if possible).
Similar threads
- Replies
- 258
- Views
- 13K
- Replies
- 4
- Views
- 567
- Replies
- 77
- Views
- 7K


