Edit: Here is a research paper on very low states of charge. They conclude long periods of storage below 2.0V causes capacity loss.
In freight classification, lithium-ion batteries are classed as dangerous goods and are therefore subject to stringent regulations and guidelines for certification for safe transport. One such guideline is the requirement for batteries to be at a state of charge of 30%. Under such conditions, a...

www.nature.com
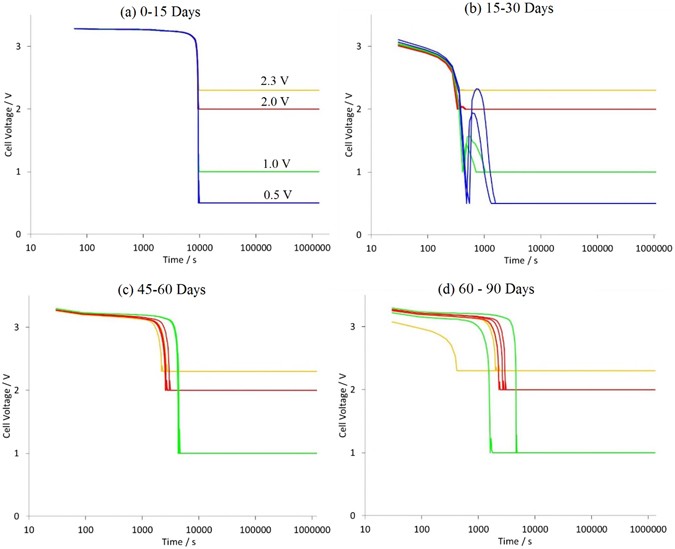
"Capacity dropped by more than 35% after 30 days of storage at 0.5 V, which posed a safety risk"
" In contrast, the 2.0 V storage results exhibited a marginal increase in storage capacity post 30 days, rising to a capacity fade of 7.1%. After 90 days of storage at 2.0 V the final capacity fade was 11.1%. Cells stored at 2.3 V exhibited capacity increase after 15 days, although within the error bar (due to cell to cell variation). However, there was a confirmed overall rise of 2.6% in cell capacity after 90 days for all three cells stored at 2.3 V."
"The Ohmic resistance (Ro) includes both ionic resistance of the electrolyte and electronic resistance of the electrode and current collectors
28,
29,
30,
31. Under low voltage storage, it is reported that the copper current collector reacts with electrolyte components resulting in corrosion
16,
17,
18, leading to higher Ro"
"More specifically, when cells are stored at low voltages over an extended period, the copper current collector attached to the carbon electrode is oxidised to Cu2+ and dissolves into the electrolyte
14,
19,
32. The subsequent reduction in contact between the current collector and active electrode material manifests as an increase in Ohmic resistance. Although previous studies such as that of Jeevarajan
et al.
33 reported slight increases of cell resistance under over-discharge conditions, the onset and rate of copper dissolution is cell specific."
"Under over-discharge conditions, over-deintercalation of lithium at the negative electrode can cause decomposition of the solid electrolyte interphase (SEI). When the cell is re-charged, new SEI film forms on the graphite anode. The growth of the SEI film leads to degradation of the electrochemical charge-transfer processes at the electrode-electrolyte interface
15,
34,
35."
"Furthermore, the decomposition of SEI leads to gas generation at the negative electrode. The generation of gases, typically CO2 and CO, cause swelling within the cell
14 and consequently a resistance rise due to electrical contact loss thorough delamination."